- Blog
- Mkvtools for mac torrent
- Actions server minecraft
- Www akila gujarati news paper
- Density water
- Adventure capitalist codes for gold
- Video poker by pokerist
- Wetter esse n
- Bitnami mean stack environment
- Royal order garter
- Revo america
- Troy kipper
- Easy numbering for pc
- Flac to mp3 320
- Cartoons logo quiz answers
- Chapter 7 we were here together ladder
- Mac rss reader newsfire
- May 2016 scrivener better than writeitnow 5
- Spotify sign in
- Surface bobok 2
- R sublime merge
- Best deck for arena 5
- Giving preferential treatment makes equal treatment unequal
- Zombie survival games free download
- Medusa skates
- Secrets of the lost tomb second edition
- Uninstall amsn
- Mozilla firefox browser free
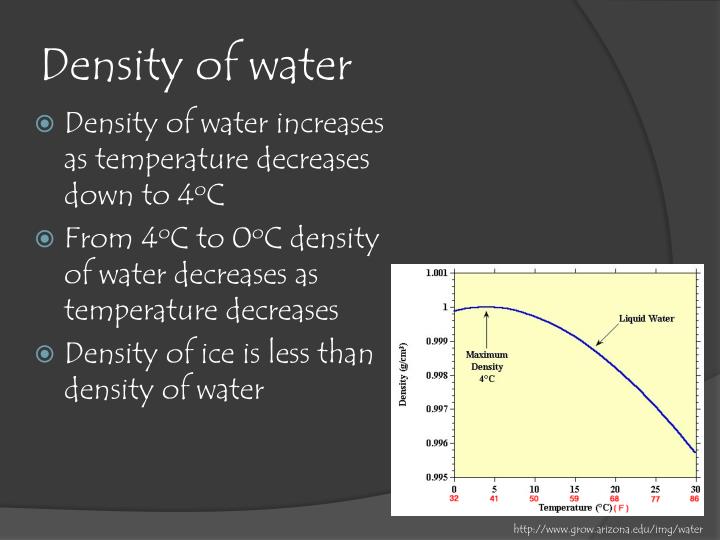
This prevents atmospheric oxygen from reaching the bottom waters. In some cases, such as happened at Ice Lake in April, 19, the surface water may warm up rapidly immediately after ice-out, causing the lake to stratify thermally without completely mixing. However, if allowed to stand undisturbed, the more buoyant (less dense) oil will float to the top and a two-layer system will develop. To overcome the buoyant force, so the two fluids can be uniformly mixed together. When you shake it up you are supplying the energy The oil is lighter (more buoyant) than the vinegar which is mostly water. Imagine a bottle of salad dressing containing vegetable oil and vinegar. It is useful to visualize a more extreme example of density stratification. As Figureģ suggests, the relatively large differences in density at higher temperaturesĪre very effective at preventing mixing. For a while winds may still mix the lakeįrom bottom to top, but eventually the upper water becomes too warmĪnd too buoyant to mix completely with the denser deeper water. As the temperature rises, the water becomes This isĪfter this spring turnover, the surface water continues The temperature (density) of the surface water equals the bottom water, very little wind energy is needed to mix the lake completely. Water above that layer will be cooler, approachingĠ☌ just under the ice. In lakes of the upper Midwest and at higher elevations, the water near a lakesīottom will usually be at 4☌ just before the lake´s ice cover melts in the spring. Because of this density-temperature relationship, many lakes in temperateĬlimates tend to stratify, that is, they separate into distinct layers. The density/temperature relationship of fresh water is shown in Figureģ. Water, in contrast, is most denseĪt 4☌ and becomes less dense at both higher and lower temperatures. The molecules become more tightly packed and consequently the compound As most compounds change from a liquid to a solid, Consequently ice floats, while water at temperatures justĪbove freezing sinks. However, density,Īnother physical characteristic of water, plays an important role inįrom most other compounds because it is less dense as a solid than asĪ liquid. Section), decreasing exponentially with depth. With depth might be expected to resemble Figure 2 (see the Light In the absence of wind, a temperature profile The intense sunlight of spring is absorbed in the waterĬolumn, which also heats up as the average daily temperature of theĪir increases. These measurements are often made with a CTD instrument, where the instrument is placed in the ocean water from a ship or a platform.With depth. You just need to measure the salinity, temperature and pressure to be able to find density. Density is usually calculated using an equation. If you wanted to measure the density of ocean water, you would have to collect a sample of sea water and bring it back to the laboratory to be measured. The density of ocean water is rarely measured directly. That is, water moves along the layers with the same density. Circulation in the depths of the ocean is horizontal. The deep ocean is layered with the densest water on bottom and the lightest water on top. So, the density of ocean water increases and increases as you go to the bottom of the ocean. The temperature of the ocean decreases and decreases as you go to the bottom of the ocean. So a layer of water with higher salinity can actual float on top of water with lower salinity if the layer with higher salinity is quite a bit warmer than the lower salinity layer. There is one catch though! Temperature has a greater effect on the density of water than salinity does. Given two layers of water with the same salinity, the warmer water will float on top of the colder water. Less dense water floats on top of more dense water. Increasing salinity also increases the density of sea water. So, the colder the water, the more dense it is. Ocean water gets more dense as temperature goes down. There are two main factors that make ocean water more or less dense than about 1027 kg/m 3: the temperature of the water and the salinity of the water.
Density of ocean water at the sea surface is about 1027 kg/m 3. Ocean water is more dense because of the salt in it.

The density of pure water is 1000 kg/m 3. A fun activity dealing with the density of salt water versus fresh water
- Blog
- Mkvtools for mac torrent
- Actions server minecraft
- Www akila gujarati news paper
- Density water
- Adventure capitalist codes for gold
- Video poker by pokerist
- Wetter esse n
- Bitnami mean stack environment
- Royal order garter
- Revo america
- Troy kipper
- Easy numbering for pc
- Flac to mp3 320
- Cartoons logo quiz answers
- Chapter 7 we were here together ladder
- Mac rss reader newsfire
- May 2016 scrivener better than writeitnow 5
- Spotify sign in
- Surface bobok 2
- R sublime merge
- Best deck for arena 5
- Giving preferential treatment makes equal treatment unequal
- Zombie survival games free download
- Medusa skates
- Secrets of the lost tomb second edition
- Uninstall amsn
- Mozilla firefox browser free
